Methods for plastic surgery of a bone defect of the skull
A bone defect in the skull is a hole in it that forms after neurosurgical intervention on the organs of the central nervous system or as a result of trauma.
Let's talk in more detail about skull defects in medical practice. After removing a section of bone and creating a hole in the skull, patients experience not only psychological discomfort, but also the so-called “trepanned skull syndrome.” In both cases, the hole must be closed with an implant. And now about everything in more detail.
Medication regimen
After surgical treatment, a sterile bandage and a special patch are applied to the operated eye. The next day they are removed and drug therapy is started. First, moisten a cotton pad in chloramphenicol drops (0.25%) and wipe the closed eyelid with it. Open the eye and instill Levomycetin according to the instructions and recommendations of the ophthalmologist.
The classic therapeutic regimen in the postoperative period after cataract removal includes the following combination of drugs:
- Antibacterial drops in combination with the glucocorticoid Dexamethasone.
Doctors prescribe these medications to prevent possible infection during the postoperative period. They help relieve inflammation, swelling, redness, and soothe the eye. They are instilled 6 times a day, at regular intervals. Then another 3 weeks 4 times. If it is difficult to remember your medication regimen, you can make yourself a reminder.
- Non-steroidal anti-inflammatory drops.
This group of drugs is used to relieve pain, to prevent the development of complications after cataract removal, for example, swelling of the macula, and elimination of inflammatory processes. The prescribed course is a month. They are buried according to different schemes. Diclofenac is used 4-6 times a day, and the modern pharmacological agent Broxinac only 1 time. NSAIDs are used until the immune system copes with the so-called sterile aseptic inflammation.
- Gels and moisturizing drops.
Gels are necessary to speed up the healing of the cornea. For unpleasant sensations in the eye (foreign body sensation), moisturizing drops are recommended.
The recovery period lasts differently for different people. This depends on age, the state of immunity, the presence of chronic diseases that weaken the body and inhibit recovery.
RECOMMENDATION! The main thing that should not be done after eye cataract removal is to ignore the recommendations of the attending physician. It is prohibited to independently change the scheme of use of medicinal drops and the amount of instilled drugs. At least 15 minutes should pass between the first and next instillation.
Consequences of skull defects
The bones of the skull not only protect the brain from mechanical damage. The absence of one area can cause a lot of unpleasant symptoms or complications for the patient. It is their presence that serves as a direct indication for surgical plastic surgery.
It could be:
- Headache. It occurs not only at the site of the defect, but also affects other areas. Most often, the symptom intensifies or occurs when there is a change in atmospheric pressure and air temperature. The so-called meteosensitivity appears.
- "Astheno-neurotic syndrome." These include complaints of absent-mindedness, decreased concentration, difficulties in solving intellectual problems, frequent mood swings, decreased ability to withstand stress and tolerance to alcohol.
- Protrusion of the contents of the skull outward. When coughing or sneezing, physical stress or exertion, part of the brain substance can protrude through an opening in the skull, under the skin. This leads to traumatization of internal tissues at the edges of the bone defect.
- Symptomatic epilepsy.
- Mental disorders.
NSICU.RU neurosurgical intensive care unit website of the intensive care unit of the N.N. Research Institute Burdenko
After removal of brain tumors, brain edema may develop and intracranial pressure (ICP) may increase. One of the probable and least studied reasons for this is a violation of venous outflow. The severity of clinical manifestations and outcomes of venous discirculation vary significantly from headache and nausea to pre-comatose state and death [14,17,22,26]. The variability of the clinical picture is determined by the number of segments of the venous system in which outflow disturbance occurs. This was shown in the experiment of Fries G et al. [14], when pigs were sequentially occluded from the superior sagittal sinus, pontine and cortical veins, while assessing the clinical condition of the animals, measuring ICP and water content in the brain tissue. It was found that severe cerebral edema, intracranial hypertension (ICH), destruction of the blood-brain barrier, and then arterial hypoperfusion leading to cerebral infarction developed only with simultaneous occlusion of the sinus, bridging and cortical veins, when both retrograde and collateral venous blood flow ceased [1, 14]. At the same time, it has been established that the most pronounced neurological deficit occurs when the outflow of blood to the deep and parasagittal veins is impaired or when a large number of the Venusylvian group is involved [24].
There are currently no generally accepted protocols for the correction of ICH that has developed as a result of impaired venous outflow. We present a clinical case of a patient with meningioma of the wing of the sphenoid bone, who in the early postoperative period acutely developed impaired venous outflow and persistent ICH.
Clinical observation.
Patient Ch., 47 years old, was admitted to the Institute with a diagnosis of “Tumor of the base of the middle cranial fossa on the left.” Upon admission, a decrease in visual acuity on the left was detected - counting fingers in the nasal part of the visual field, left-sided exophthalmos and signs of an effect on the left oculomotor nerve. An MRI examination of the brain revealed a meningioma at the base of the middle cranial fossa on the left (Figure 1).
An operation was performed - subtotal removal of meningioma of the medial parts of the base of the middle cranial fossa on the left using a pterional approach. The cavernous sinus was infiltrated by the tumor. This part of the tumor was not removed. Two large veins of the Sylvian group that had grown into the tumor capsule were coagulated intraoperatively.
Blood loss did not exceed 700 ml and was adequately replaced.
Awakening from anesthesia sleep was at the usual time. There was no increase in cerebral and focal neurological symptoms compared to the preoperative level.
The patient was extubated 2 hours after surgery. Hemodynamics were stable.
Homeostatic parameters were within normal limits.
The condition worsened sharply 12 hours after the operation. A coma and right-sided hemiparesis developed (3 – 4 points). Due to respiratory failure, the patient was intubated and mechanical ventilation was started in the SIMV+PS mode. A CT examination of the brain revealed predominantly left hemispheric edema, a massive focus of low density located in the fronto-parietal-temporal region on the left, and a displacement of the midline structures from left to right by 9 mm. The basal cisterns were not visualized, and the ventricular system was compressed (Fig. 2). Transcranial Doppler sonography showed linear blood flow velocity within normal limits. A subdural/parenchymal ICP sensor (Codman, USA) was installed. Severe ICH was detected (ICP 35 – 45 mmHg).
To correct ICH, options in the treatment protocol for patients with traumatic brain injury were used, since there are generally accepted protocols for correcting ICH when the venous drainage system is disrupted. The head end of the bed was raised 30º. Moderate hyperventilation, sedation with propofol (45 mcg/kg/min), analgesia with fentanyl (0.02 mcg/kg/min), and muscle relaxation with pipecuronium (0.5 mcg/kg/min) were started. These measures were ineffective - ICP remained at the level of 30 - 35 mmHg. 20 minutes after the start of therapy. After this, osmotherapy with mannitol (up to 1.5 g/kg) was used. After 10 minutes, ICP decreased to 20–25 mmHg, however, after 30 minutes, severe ICH developed again (up to 40 mmHg). Repeated mannitol infusion was ineffective.
A decision was made to proceed with moderate hypothermia. Induction of hypothermia began one hour after deterioration. External cooling and intravenous administration of cooled saline solution at a dose of 20 ml/kg were used. A temperature of 33º C was reached two hours after induction. ICP was effectively stabilized at 10–12 mmHg. During hypothermia, hypokalemia (3.1 – 3.3 mmol/l) and hypomagnesemia (0.39 – 0.41 mmol/l) developed, which were effectively corrected by the use of potassium and magnesium preparations. To prevent the development of hypocoagulation, taking into account the early postoperative period and the effects of hypothermia, plasma transfusion was performed at a dose of 15 ml/kg. At the same time, the prothrombin index was in the range of 75–85%, activated partial thromboplastin time was 28–33 seconds, fibrinogen was 3.4–3.9 g/l.
Body temperature was maintained at 33ºC for 24 hours. Before the start of warming, a control CT study of the brain was performed, which revealed positive dynamics in the form of the appearance of the basal cisterns and a decrease in the displacement of the median structures to the right to 5 mm. However, cerebral edema and a focus of low density in the left hemisphere persisted (Fig. 3).
Upon reaching 35ºC (10 hours after the start of rewarming), a clear tendency towards ICH appeared, so the rate of rewarming was reduced to ≈ 0.05 degrees per hour. Outside of sedation, the patient followed simple instructions, and the right-sided hemiparesis regressed.
Thus, already at this stage the positive effects of hypothermia were identified in the form of regression of general cerebral and focal neurological symptoms.
A temperature of 36ºC was reached after 20 hours. At this temperature, ICH developed up to 30 mmHg, resistant to sedation and osmotherapy with mannitol. This was an indication for performing external decompressive trepanation with plastic surgery of the dura mater. From the operation protocol it follows that, despite the tension of the dura mater, revealed after removal of the bone flap, the brain pulsated and was not vulnerable when touched. This was probably due to previous treatment, in particular the cerebroprotective effects of hypothermia. After dissection of the dura mater and expansion of the bone defect, the tension in the medulla decreased significantly, which made it possible to perform dura mater plastic surgery without technical difficulties. Decompression and plastic surgery of the dura mater made it possible to effectively stabilize ICP at a level of 10–15 mmHg.
Sedation continued for another 2 days after external decompression, and ICP monitoring continued for 5 days. There were no episodes of intracranial hypertension during this period. A CT examination revealed positive dynamics (Figure 4). The patient was conscious, but disoriented in place, time, and personal situation.
Movements were detected in all limbs, without clear asymmetry. Mechanical ventilation was stopped 6 days after decompressive trepanation, and after another 5 days the patient was transferred from intensive care to the neurosurgical department.
After 1.5 months, the patient’s condition was stable. Korsakov's syndrome was detected.
There were no speech or motor disturbances. An MRI study of the brain was performed with venography and in diffusion mode (Fig. 5), which revealed a lack of blood flow in the transverse and sigmoid sinuses on the left. In addition, an extensive lesion of cerebromalacia in the frontotemporal region of basal localization on the left was diagnosed, which was also visualized during a CT examination in the acute period as a focus of low density.
Analysis of clinical symptoms, CT and MRI data of the brain suggests that the cause of the formation of this lesion is an acute disturbance of venous outflow. In addition, an ischemic focus was visualized in the occipital region on the left, which has a cone-shaped shape and is located in the projection of the posterior cerebral artery basin. The most likely reason for the formation of this lesion is compression of the posterior cerebral artery on the tentorium of the cerebellum during swelling and dislocation of the brain in the acute period of the disease.
After 2.5 months, the patient developed hydrocephalus, which was an indication for lumboperitoneal shunting. After 4 months, the patient was discharged from the Institute in stable condition. The patient could care for herself independently.
The severity of Korsakoff's syndrome has decreased. 6 months after discharge, the patient is planned to be re-hospitalized to perform plastic surgery of the bone defect. The differential diagnosis for deterioration of the patient's condition was inter-arterial ischemia and impaired venous outflow. Venous disorders are characterized by: - absence of awakening from anesthesia sleep or deterioration of the condition several hours after surgery [17]; - CT examination can visualize a focus of reduced density in the first day after surgery due to hydrostatic edema [2]; - intraoperative data on damage to the veins or sinuses; - blood flow velocity during transcranial Doppler ultrasound remains normal or decreases [4,29,30,33]. - MRI venography reveals altered parameters [2]. However, performing an MRI examination in the acute period in the presence of severe ICH is fraught with the development of life-threatening complications.
So, differential diagnosis made it possible to suspect the presence of a venous outflow disorder in the patient. The pathogenesis of the developed condition can be represented as follows.
Intraoperative occlusion of the Sylvian veins included in the tumor led to local venous discirculation. The lack of blood flow through the transverse, sigmoid sinus (possibly congenital) and cavernous sinus (tumor infiltration) limited the compensatory possibilities of collateral redistribution of venous outflow. This led to a gradual increase in cerebral edema within 10–12 hours after surgery, which, in turn, caused discirculation in the system of deep veins of the brain.
This was the cause of acute deterioration 12 hours after surgery with the development of a comatose state due to severe cerebral edema and dislocation.
Currently, there is no protocol for correcting ICH in cases of impaired venous outflow.
The use of intensive care recommendations for the correction of ICH in patients with brain tumors, TBI and stroke was ineffective in our observation. Glucocorticosteroid hormones are not used for intensive treatment of cerebral edema caused by impaired venous outflow [17,26]. Raising the head of the bed, sedation, analgesia, muscle relaxation, moderate hyperventilation and osmotherapy were ineffective - ICH persisted.
If the listed options are ineffective in reducing ICP, the use of aggressive methods is indicated: barbiturate coma, external decompression or moderate hypothermia.
Barbiturates were not used because, according to the Cochrane Collaboration, which specializes in organizing and analyzing research results using the principles of evidence-based medicine, it follows that: “there is no evidence that barbiturate therapy in patients with severe traumatic brain injury improves outcomes. Barbiturates cause arterial hypotension in every fourth patient. The hypotensive effect of barbiturates will offset the positive effect of lowering ICP on cerebral perfusion pressure..." [28].
External decompressive trepanation was not used at this stage, since, according to Greenberg MS, when venous outflow is impaired due to venous thrombosis, it leads to a decrease in ICP, but does not improve disease outcomes [17]. With external decompression, intracranial relationships change, cerebrospinal fluid dynamics changes, and the risk of hemorrhage into the remnants of the tumor or the focus of ischemia, if present, increases.
It is known that with severe cerebral edema after decompression, entrapment of brain tissue in a bone defect can develop with the development of ischemia and secondary disruption of venous outflow in this area [11,27].
Moderate hypothermia (32 - 34ºС) is an effective method of controlling ICP [5,7,18], and has a cerebroprotective effect. The mechanism of cerebroprotection is to reduce the level of metabolism [6,9], reduce the permeability of the blood-brain barrier [13], reduce the concentration of excitatory amino acids and pro-inflammatory interleukins in damaged brain tissues [3,15], and reduce lipid peroxidation [19]. It is known that in experimental and clinical conditions, moderate hypothermia significantly reduces ICP, reduces the area of ischemic brain damage and can improve outcomes in a wide range of pathologies of the central nervous system: head injury, stroke, cerebral edema after cardiac arrest [10,16,20,23,25,31 ,32].
In the above observation, hypothermia during its implementation, firstly, effectively stabilized ICP, and secondly, it made it possible to protect the brain from ischemia. In contrast to oarterial ischemia, which develops, for example, due to clipping of an artery, when venous outflow is impaired, ischemia develops later. First, pronounced hydrostatic cerebral edema appears. This is precisely what manifested the deterioration of the condition in the above observation. If collateral or retrograde venous blood flow is not possible, then cerebral edema, incompatible with life, acutely develops. If collateral or retrograde venous blood flow is preserved, when its compensatory redistribution is possible, the severity of edema will vary significantly [14]. If the outflow disturbance persists, areas of the brain will form that will not receive an adequate volume of arterial blood. As a result, ischemia will develop. So, with venous infarction, hydrostatic edema first develops, and then ischemia and ischemic edema. In arterial infarction, ischemia develops first, followed by edema. Since in the above observation, when normothermia was achieved, there was regression of right-sided hemiparesis and restoration of consciousness, and subsequently there were no motor disorders, hypothermia probably had a cerebroprotective effect. In addition, the hypothermia provided adequate conditions for subsequent external decompression.
Hypothermia is an aggressive method for correcting ICH. The most frequently described complications in the literature are hypocoagulation [8,35], fluid and electrolyte disturbances [21], hemodynamic disorders [12,34] and infectious and inflammatory complications [8,34]. The most dangerous manifestations of hypocoagulation are intracranial hemorrhages. This led to the prophylactic use of fresh frozen plasma in the above observation. Fluid and electrolyte disturbances during hypothermia are manifested by hypokalemia and hypomagnesemia [21]. Hemodynamic disorders are usually manifested by sinus bradycardia. More dangerous rhythm disturbances have been described - asystole and ventricular fibrillation, however, they develop either at a temperature of less than 28ºC or with a duration of hypothermia of more than 48 hours [8,12,34]. The hypokalemia, hypomagnesemia and hemodynamically insignificant bradycardia that developed in the above observation did not threaten vital functions and were corrected immediately upon their development. Thus, moderate hypothermia has proven to be a relatively safe method of controlling ICP.
The warming period is an important stage in the management of hypothermia. In our observation, during warming, a tendency to increase ICP appeared. This is consistent with the data of a number of authors. Thus, Schwab S et al showed that warming is a period of high risk for the development of recurrent and persistent ICH, which can be fatal [31,32]. The authors proved that rewarming duration exceeding 16 hours significantly reduces mortality. Therefore, the rate of warming in our observation was ≈ 0.05º per hour. Despite this, stable ICH gradually developed, which became an indication for external decompressive trephination with dural plastic surgery. The chosen tactics made it possible to prevent increased brain dislocation and to preserve the structure of the brain parenchyma at the time of decompression.
Literature data and our observation indicate that hypothermia, due to its cerebroprotective effect, can reduce the area of an already formed ischemic focus and prevent further ischemic damage. However, during the warming period, the risk of developing recurrent resistant ICH remains. Under these conditions, it is necessary to perform external decompression with plastic surgery of the dura mater.
Hypothermia in this case will create more favorable conditions for performing the operation.
In conclusion, it must be said that in patients with tumors of basal localization in the early postoperative period, cerebral edema may develop due to impaired venous outflow. At the same time, stable ICH develops rapidly. It is quite difficult to differentiate impaired venous outflow from other possible causes of the development of cerebral edema in the acute period. However, this condition should always be considered as a possible cause for the development of persistent ICH. Moderate hypothermia is an effective and safe method for correcting cerebral edema and ICH in cases of impaired venous outflow. Intracranial hypertension that develops during the warming stage is an indication for external decompression. And, of course, further research is needed in this interesting and promising direction.
Literature
- Bekov D.B., Mikhailov S.S. “Atlas of arteries and veins of the human brain,” M. Medicine, 1979, pp. 95-96.
- Kornienko V.N., Pronin I.N. Diagnostic neuroradiology. M. Publishing house Andreeva T.M., 1368 pp. 2007.
- Aibiki M, Maekawa S, Ogura S, et al: Effects of moderate hypothermia on systemic and internal jugular plasma IL-6 levels after traumatic brain injury in humans. J Neurotrauma 1999; 16:225-232.
- Alexandrov A, Joseph M: Transcranial Doppler: An Overview of its Clinical Applications. The Internet Journal of Emergency and Intensive Care Medicine 2000; Vol. 4, No. 1 (ISSN: 1092–4051).
- Bayir H, Clark RSB, Kochanek PM Promising strategies to minimize secondary brain injury after head trauma. Crit Care Med 2003; 31:S112-S117.
- Berntman L, Welsh FA, Harp JR: Cerebral protective effect of low-grate hypothermia. Anesthesiology 1981; 55:495-498.
- Bernard SA, Buist M Induced hypothermia in critical care medicine: A review. Crit Care Med 2003; 31:2041–2051.
- Bernard S.A. Therapeutic hypothermia after cardiac arrest: Now a standard of care. Crit Care Med 2006; Vol. 34 P 923-924.
- Chopp M, Knight R, Tidwell CD, et al: The metabolic effects of mild hypothermia on global cerebral ischemia and recalculation in the cat: Comparison to normothermia and hyperthermia. J Cereb Blood Flow Metab 1989; 9:141-148.
- Colbourne F, Sutherland G, Gorbett D: Postischemic hypothermia: A critical appraisal with implications for clinical treatment. Mol Neurobiol 1997; 14:171-201.
- Csokay A, Pataki G, Nagy L, Belan K. Vascular tunnel construction in the treatment of severe brain swelling caused by trauma and SAH. (Evidence based on intra-operative blood flow measure). Neurological research 2002; 24:157-160.
- Danzl DF, Pozos RS: Accidental hypothermia. N Engl J Med 1994; 331:1756–1760.
- Dietrich WD, Busto R, Halley M, et al: The importance of brain temperature in alterations of the blood-brain barrier following cerebral ischemia. J Neuropathol Exp Neurol 1990; 49:486-497.
- Fries G, Wallenfang T, Hennen J, et al Occlusion of pig superior sagittal sinus, bridging and cortical veins: multistep evolution of sinus-vein thrombosis. J. Neurosurg. 1992; 77:127-133.
- Globus MY, Alonso O, Dietrich WD, et al: Glutamate release and free radical production following brain injury: Effects of posttraumatic hypothermia. J Neurochem 1995; 65:1704–1711.
- Goto Y, Kassell NF, Hiramatsu K, et al: Effects of intraischemic hypothermia on cerebral damage in a model of reversible focal ischemia. Neurosurgery 1993; 32:980-984.
- Greenberg MS. Handbook of neurosurgery. pp 609,611,891. Thieme, 2001.
- Hofmeijer J, van der Worp H, Kappelle LJ Treatment of space-occurring cerebral infarction. Crit Care Med 2003; 31:617-625.
- Karibe H, Chen SF, Zarow GJ, et al: Mild intraischemic hypothermia suppress consumption of endogenous antioxidants after temporary focal ischemia in rats. Brain Res 1994; 649:12-18.
- Kawai N, Okauchi M, Morisaki K, et al: Effects of delayed intraischemic and postischemic on a focal model of transient cerebral ischemia in rats. Strike 2000; 31:232-239.
- Koht A, Cane R, Cerullo LJ: Serum potassium levels during prolonged hypothermia. Int Care Med 1983; 9:275 – 277.
- Kurita H, Shin M, Ueki K, Kawamoto S, Kirino T. Congestive brain oedema associated with a pial arteriovenous malformation with impaired venous drainage. Acta Neurochir. 2001; 143:339-342.
- Maier CM, Sun GH, Kunis D, et al: Delayed induction and long-term effects of mild hypothermia in a focal model of transient cerebral ischemia: Neurological outcome and infarct size. J Neurosurg. 2001; 94:90-96.
- McElveen WA, Gonzales RF, Keegan AP: Cerebral venous thrombosis. E-medicine (https://www.emedicine.com/neuro/topic642.htm), 2006.
- Nolan JP, Morley PT, Hoek TL, et al: Therapeutic hypothermia after cardiac arrest. An advisory statement by the Advancement Life supporting Task Force of the International Liaison Committee on Resuscitation. Resuscitation 2003; 57:231-235.
- Park JH, Yoon SH New concept of cerebrospinal fluid dynamics in cerebral venous sinus thombosis. Medical Hypotheses 2007, article in press.
- Polin RS, Shaffrey ME, Bogaev CA. Decompressive bifrontal craniectomy in the treatment of severe post-traumatic cerebral edema. Neurosurgery 1997; 41:84-92.
- Roberts I. Barbiturates for acute traumatic brain injury. Cochrane Database of Systematic Reviews 2005(2):CD000033.
- Saqqur M, Zygun D, Demchuk A: Role of transcranial Doppler in neurocritical care. Crit Care Med 2007; 35(5) Suppl May: S216-S223.
- Schreiber SJ, Stolz E, Valdueza JM: Transcranial ultrasonography of cerebral veins and sinuses. Eur J Ultrasound 2002 Nov; 16(1-2): 59-72.
- Schwab S, Georgiadis D, Berrouschot J, et al: Feasibility and safety of moderate hypothermia after massive hemispheric infarction. Stroke 2001; 32:2033–2035.
- Schwab S, Schwarz S, Spranger M, et al: Moderate hypothermia in the treatment of patients with severe middle cerebral artery infarction. Stroke 1998; 29:2461–2466.
- Stolz E, Kaps M, Kern A, et al.: Transcranial color-coded duplex sonography of intracranial veins and sinuses in adults. Reference data from 130 volunteers. Stroke 1999 May;30(5): 1070 – 1075.
- The hypothermia after cardiac arrest study group: mild therapeutic hypothermia to improve the neurological outcome after cardiac arrest. New Engl J Med 2002; 346:549-556.
- Valeri CR, MacGregor H, Cassidy G, et al: Effects of temperature on bleeding time and clotting time in normal male and female volunteers. Crit Care Med 1995; 23:698-704.
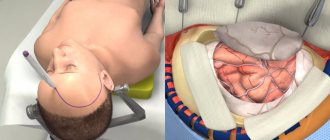
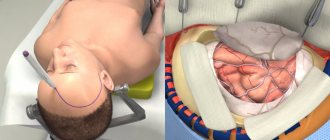
Cranioplasty
This is the name of the operation to restore the tightness of the skull. It is designed to restore the brain’s protection from external influences, normalize blood and lymph flow, eliminate the above symptoms, cosmetic defects and relieve psychological discomfort.
The essence of the procedure is to close the hole in the skull with an implant. A competent neurosurgeon will help you understand the great variety of implants offered by medicine today and choose the best one. Fill out an application on the website or call us to get advice from a leading specialist to choose the right implant and discuss the features of the upcoming operation.
We will tell you what types of cranioplasty implants there are and how the operation to install them is performed.
Complications after surgery
Surgical manipulations on the brain are always associated with a high risk of postoperative complications. Complications can occur both during and after the operation.
Infectious
If the rules of asepsis are observed during manipulations, infectious complications are minimized. Their risk increases with traumatic brain injuries with contamination of the wound surface. The risk of developing inflammatory processes in internal organs increases with impaired mobility after surgery, in the case of stagnant processes. To reduce the risk of infection after trepanation, antibiotic therapy is administered and therapeutic exercises are prescribed.
Bleeding
The brain has an extensive blood supply. If blood vessels are damaged, the postoperative wound may bleed for 2-3 days. Blood can accumulate in the skull, permeating the brain structures, compressing the nerves. This can lead to disruption of the vasomotor and respiratory centers, and convulsions. In order to prevent these complications, drains are placed in the wound to drain fluid from the wound. If bleeding progresses, repeat surgery may be required.
Implants
There is a list of mandatory qualities that they must have:
- sufficient strength;
- biocompatibility;
- ease;
- plastic;
- do not cause infectious complications;
- do not have a carcinogenic effect.
Implants can be made from the patient's own bone (autografts) or artificial (xenografts).
Autografts
This is bone that has been temporarily removed to access the patient's brain tissue. If the bone needs to be preserved to close a skull defect later, it is placed under the skin of the patient's abdomen or anterior outer thigh. If such preservation is not possible, other techniques using the person’s own bone tissue are used. And if it is impossible to replace the defect with autografts, they resort to materials replacing them.
Xenografts
The so-called “artificial bones” are made of metal (aluminum or titanium), using methyl methacrylate or hydroxyapatite.
Each of the mentioned implants has its own advantages and disadvantages. For example, hydroxyapatite can be used for defects up to 30 square centimeters. This material is completely biocompatible. And it is able to turn into its own bone tissue after 18 months with small defects. And the risk of infectious complications for this type of implant is the lowest among similar ones.
Titanium is the least likely to cause local inflammatory reactions. And from it it is possible to recreate lost fragments of the facial skeleton, the base of the skull, the vault and the walls of the orbit. This is done using simulation and neuronavigation in the preoperative period.
The doctor will help you understand the variety of implants, as well as find out what the latest medical developments in the field of implantology are, during an in-person consultation. Fill out an application on our website and we will make an appointment with a leading specialist in the field of neurosurgery.
In each specific case, it is important to take into account not only the direct benefits for a person, but also the aesthetic and psychological aspects. As well as restrictions imposed on the patient in connection with the choice (for example, a metal implant does not allow the patient to subsequently resort to MRI examination).
Early postoperative period
To avoid hematoma, rubber tubes are placed under the flaps, the ends of which remain under a protective bandage. Blood flows through the tubes, soaking the bandage. If the bandage gets significantly wet, it is not changed, and a new bandage is additionally wound on top. If, at the end of the operation, the meninges are not completely sealed, then traces of cerebrospinal fluid may appear in the leaking blood mass.
The outlet tubes are usually removed one day after surgery is completed. To prevent leakage of cerebrospinal fluid and eliminate the risk of infection through the areas where graduates were located, provisional or additional sutures are placed and tied.
On the first day after surgery, it is necessary to monitor the condition of the bandage in the trepanation area. Significant swelling of the bandages over the operated area is due to postoperative hematoma, which can cause a rapid increase in swelling of the soft tissues of the forehead and eyelids, and bleeding in the orbital area. A very dangerous consequence that appears at an early stage after craniotomy can be secondary liquorrhea, which can provoke infection of the cranial contents, causing meningitis and encephalitis. In this regard, it is extremely important to promptly detect the presence of light liquid in the blood mass soaking the bandage and take urgent measures.